May Bulletin
Issue 116
Community Notices
Next Marble Center Seminar with the Anderson Lab, Monday May 18th (4-5pm, Luria Auditorium)
Next Marble Center seminar is on Monday May 18th (4-5pm) at the KI Luria Auditorium with a research talk by Dr. Yuang Chen of the Anderson lab. The title of his talk is “In vivo CAR NK Cell Engineering via Ligand-directed LNPs for Immunotherapy.”
Following the talk, we will be hosting a hot topic presentation by Dr. Mushriq Al-Jazrawe, scientific director of the High Throughput Sciences (HTS) facility in the Swanson Biotechnology Center at MIT's Koch Institute. The title of his talk is “The HTS Platform for Automated and Data-Driven Experimentation in Cancer Therapeutics.”
REGISTRATION OPEN: Koch Institute Annual Symposium, June 18, 2026
The 2026 Koch Institute Annual Symposium on “Diet and Metabolism in Cancer” will feature international experts from academia and leading clinical institutions and covering topics such as the influence of diet on health and cancer, diet-gut interactions, diet and the immune system, as well as translational applications for diet in anticancer therapy.
News
Thank you all for attending our Marble Center 10 Year Anniversary Celebration!
Please enjoy a selection of event photos and see if you can spot a few alumni! A video of the event will be shared soon!











































Tuning the immune response to mRNA vaccines
(News & Views by the Anderson Lab, Nature Biotechnology) Messenger RNA (mRNA) vaccines have emerged as a transformative technology for vaccination and immunotherapy. Despite their clinical success, the cellular mechanisms that underpin immune responses to mRNA vaccines remain incompletely understood. In new work in Nature Biotechnology, Marks et al. study this question in mice by selectively silencing antigen expression in specific cell types, including dendritic cells, muscle cells and hepatocytes, through incorporation of microRNA (miRNA) signals into the mRNA sequence. They find that direct antigen production in antigen-presenting cells (APCs) is not required to generate a robust immune response and that antigen expression in non-immune cells, particularly muscle, influences the immune response. The study offers intriguing insight into how antigen expression and processing shape the protection afforded by an mRNA vaccine and suggests avenues for tuning immune responses to mRNA therapeutics.
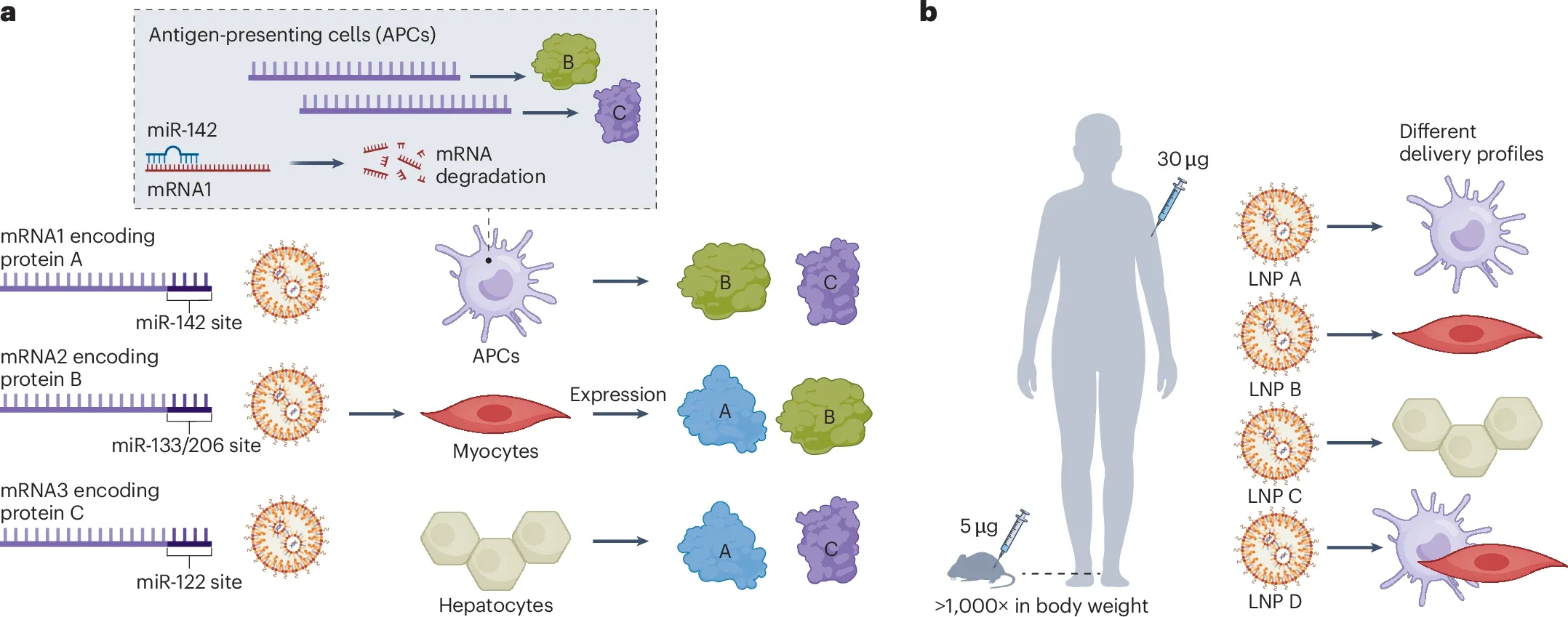
microRNA platform for cell-specific mRNA silencing and limitations in translating to humans
a. The miRNAs evaluated for silencing of mRNA translation in dendritic cells, hepatocytes and myocytes to probe the roles of antigen expression in each of these cells in the immunogenicity of mRNA vaccines. b. While a human has orders of magnitudes of higher body weight than a mouse, the dose of mRNA vaccines used in human and in mouse was not very different. Conversely, different LNP formulations may have different delivery profiles in vivo, generating distinct immune responses. Both factors should be considered, and thus caution is needed when generalizing findings with a specific LNP formulation in mice to commercially available mRNA vaccines in human.
Eli Lilly Secures In Vivo CAR-T Platform Through $7 Billion Kelonia Acquisition, Advancing Major Strategic Push Into Hematologic Oncology
(OncoDaily) Eli Lilly and Company has announced a definitive agreement to acquire Kelonia Therapeutics for up to $7 billion in cash, securing access to what may represent the most commercially viable approach yet to delivering CAR-T cell therapy at scale. The transaction, structured as a $3.25 billion upfront payment with additional milestone-based consideration, positions Lilly to compete directly in a therapeutic modality that has transformed outcomes in hematologic malignancies but has remained constrained by manufacturing complexity, cost, and accessibility. For Lilly, a company whose recent dominance has been defined by its GLP-1 franchise, the Kelonia acquisition represents a continuation of an aggressive strategic pivot toward establishing credible positions across high-value oncology segments.
Kelonia Therapeutics is a clinical-stage biotechnology company built around a proprietary in vivo gene delivery platform designed to reprogram patients’ T-cells directly within the body to recognize and attack cancer cells [The lead drug (KLN-1010) is based on a vector first envisioned by Michael Birnbaum]. This approach stands in contrast to the prevailing ex vivo CAR-T methodology, which requires harvesting a patient’s cells, engineering them in specialized manufacturing facilities, and reinfusing them following lymphodepleting chemotherapy, a process that can take weeks, cost hundreds of thousands of dollars, and is available only at a limited number of academic medical centers with the infrastructure to manage the logistical and clinical complexity. Read more…
The U.S. National Nanotechnology Initiative publishes the Supplement to the President’s 2026 Budget
This document is a product of the Committee on Technology, Subcommittee on Nanoscale Science, Engineering, and Technology of the National Science and Technology Council. It is a supplement to the President’s 2026 Budget request submitted to Congress on May 2, 2025, and serves as the Annual Report for the National Nanotechnology Initiative called for under the provisions of the 21st Century Nanotechnology Research and Development Act (15 USC §7501–7509).
Job opportunities
Research Scientist, Center for Biomedical Innovation, MIT. The Center for Biomedical Innovation (CBI) , part of the Sociotechnical Systems Research Center (SSRC), will support an ARPA-H–funded program developing a novel platform for the continuous manufacturing of extracellular vesicles (EVs); design and execute experimental workflows to develop next-generation sequencing (NGS)–based approaches for rapid detection of AA genetic material in both process and product samples. Responsibilities include conducting validation studies, such as spiking experiments, to assess sensitivity, specificity, and robustness, and integrating validated analytical tools into continuous manufacturing processes. The role also involves collaboration with interdisciplinary teams across bioprocessing, analytics, and bioinformatics to support platform development. This role focuses on advancing methods for detecting adventitious agents (AAs), including microbial and viral contaminants, within continuous EV manufacturing systems.
Business Development Manager, Wyss Institute at Harvard University. Reporting to the Senior Business Development Director, Commercialization, the Business Development Manager will provide high-level support for the Institute's fast-paced business development activities by supporting startup formation, sponsored research and collaborations, licensing, and alliance management.
Funding opportunities
| Innovative Research in Cancer Nanotechnology | May 5, 2026 | MIT HEALS Seed Grant Letter of Intent Deadline | June 1, 2026 |